Mariano Gorrachategui
Animal nutrition consultant and president of CESFAC
Co-occurrence of mycotoxins
We know that a certain mycotoxin can be produced by several types of fungi and that the same toxigenic fungus can produce several mycotoxins.
Moreover, nowadays there are different sources of raw materials. Cereals, plant protein crops, and oilseeds grow and are harvested in a diverse range of climatological conditions, determining the growth of the fungi and the toxins produced. The storage and transport conditions also affect the fungus and toxins that can be produced during that time.
Under these conditions, it’s easy to understand that the odds of finding only one mycotoxin contaminating raw materials or feed are slim. Thus, the term “co-occurrence” is increasingly being used when referring to the simultaneous presence of two or more mycotoxins in the same sample.

There are many publications in literature that highlight the co-occurrence of mycotoxins:
In Germany, Goertz et al. (2010) found corn to be simultaneously contaminated with at least 14 Fusarium mycotoxins:
- ⇰ Deoxynivalenol (DON) and its acetylated forms
⇰ Zearalenone (ZEA) - ⇰ Moniliformin (MON)
- ⇰ Beauvericin (BEA)
- ⇰ Nivalenol (NIV)
- ⇰ Eniantins (ENNs)
- ⇰ Fumonisins (FBs)
- ⇰ HT2 Toxin
En un seguimiento global, Streit y col. (2013a) indicaron que, en piensos y materias primas, el 72% de las muestras contenían más de una micotoxina.
Streit et al.., (2013b) observed 83 samples of feed and raw materials to be contaminated with 7 to 69 mycotoxins per sample, having detected up to 169 different compounds.
Other studies have revealed the simultaneous presence of several mycotoxins in samples from European countries (Almeida et al., 2011; Blajet-Kosicka et al., 2014; Driehuis et al., 2008; Labuda et al., 2005a, 2005b; Monbaliu et al., 2010), finding a high percentage of samples to be contaminated with trichothecenes (DON, AcDon, T2, HT2) and FBs at the same time, as well as with ZEA in many cases.
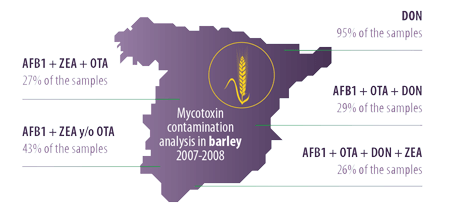
In Spain, the study published by Ibáñez-Bea et al. (2012) regarding the simultaneous presence of AFB1, ZEA, and OTA in samples of barley harvested in 2007 and 2008, revealed:
- ⇰ The presence of the three mycotoxins in 27% of the samples
- ⇰ The presence of AFB1, together with any one of the other two in 43% of the samples
The same authors detected DON in 95% of the samples and two mycotoxins from this group in 43% of the samples.
Taken together, the results of these studies reveal that 96% of the samples contained three or more mycotoxins. The most frequent combinations are:
- ⇰ AFB1, OTA, and DON in 29% of the samples
- ⇰ AFB1, OTA, DON, and ZEA en 26% of the samples
More recent data (Biomin, 2017) from the analysis of 1.378 samples indicated that 94% of the samples contained more than 10 mycotoxins and metabolites and that the average was of 28 mycotoxins.
In another study, Raj et al. (2017), after screening 113 samples of maize from Serbia and Bosnia and Herzegovina for mycotoxin contamination, found 28% of the samples to be contaminated with more than one mycotoxin. In another study, the authors reported 76% samples contaminated with one or more mycotoxins (Raj et al., 2019).
Regarding fodder, recently there has also been data published (Panasiuk et al., 2019).








 Micotoxicosis prevention
Micotoxicosis prevention