Jog Raj1*, Panagiotis Tassis2, Vasilios Tsiouris2 and Marko Vasiljević1
1Patent Co, DOO., Misicevo, Serbia
2School of Veterinary Medicine, Aristotle University of Thessaloniki, Greece.
*[email protected]
 The aim of this study was to document the efficacy of mycotoxin absorbent Minazel® Plus (MP) (modified clinoptilolite) on broiler chickens ingesting feed containing AFB1 and OTA from the 1st day up to 42nd day of age, registering alterations in health status and performance parameters. The aim of this study was to document the efficacy of mycotoxin absorbent Minazel® Plus (MP) (modified clinoptilolite) on broiler chickens ingesting feed containing AFB1 and OTA from the 1st day up to 42nd day of age, registering alterations in health status and performance parameters. |

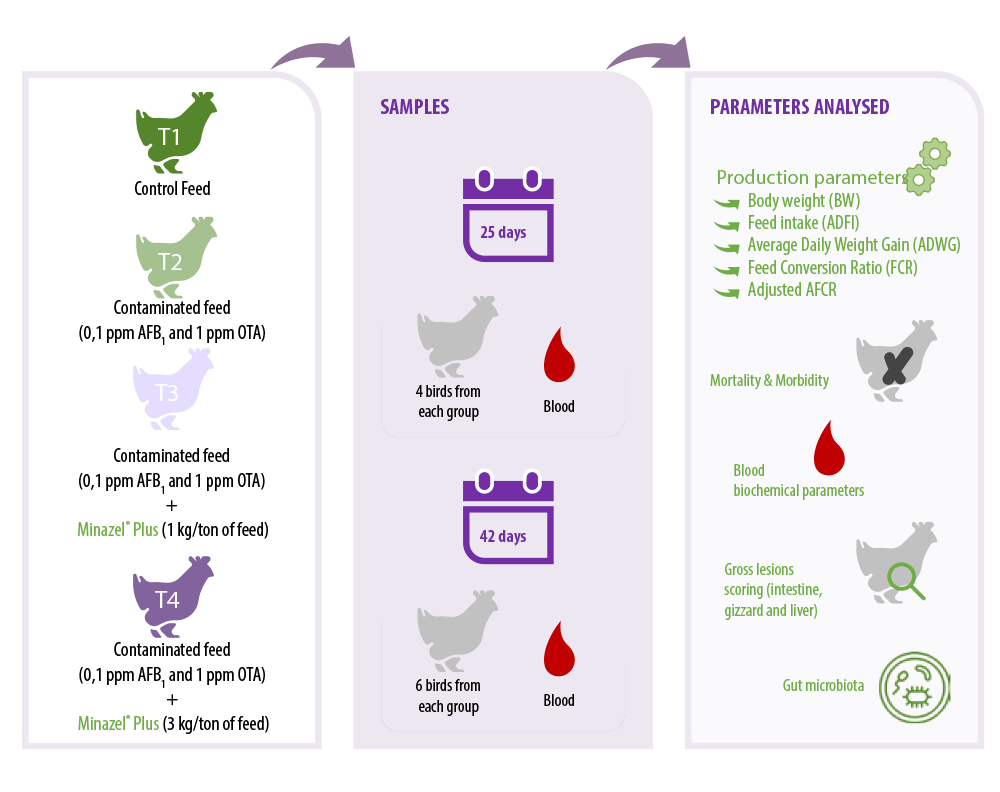
The trial was performed in the Unit of Avian Medicine experimental unit, at the Farm Animals Clinic, of the School of Veterinary Medicine of the Aristotle University of Thessaloniki, Greece in 2019. The duration of the experiment was 42 days.
The treatment groups were:
 T1: negative control group fed with control feed.
T1: negative control group fed with control feed. T2: Positive control group receiving control feed contaminated with 0.1 ppm AFB1 and 1 ppm OTA.
T2: Positive control group receiving control feed contaminated with 0.1 ppm AFB1 and 1 ppm OTA. T3: Group receiving the T2 group feed (0.1 ppm AFB1 and 1 ppm OTA) with the addition of Minazel® Plus (1 kg/ton of feed).
T3: Group receiving the T2 group feed (0.1 ppm AFB1 and 1 ppm OTA) with the addition of Minazel® Plus (1 kg/ton of feed). T4: Group receiving the T2 group feed (0.1 ppm AFB1 and 1 ppm OTA) with the addition of Minazel® Plus (3 kg/ton of feed).
T4: Group receiving the T2 group feed (0.1 ppm AFB1 and 1 ppm OTA) with the addition of Minazel® Plus (3 kg/ton of feed).
On the 25th and 42nd day of age 4 and 6 birds, respectively, with 4 replicates (from each experimental group) per sampling day were blood sampled and removed.

The following parameters were calculated: body weight (BW), feed intake (ADFI), average daily weight gain (ADWG), feed conversion ratio (FCR), as well as adjusted AFCR that was calculated based on a standard reference weight of 2,500g.
⇰ Furthermore, mortality and morbidity were investigated, as well as blood biochemical parameters tests, and gross lesions scoring of the intestine, gizzard and liver were performed. Additionally, investigation of gut microbiota was also performed.
RESULTS
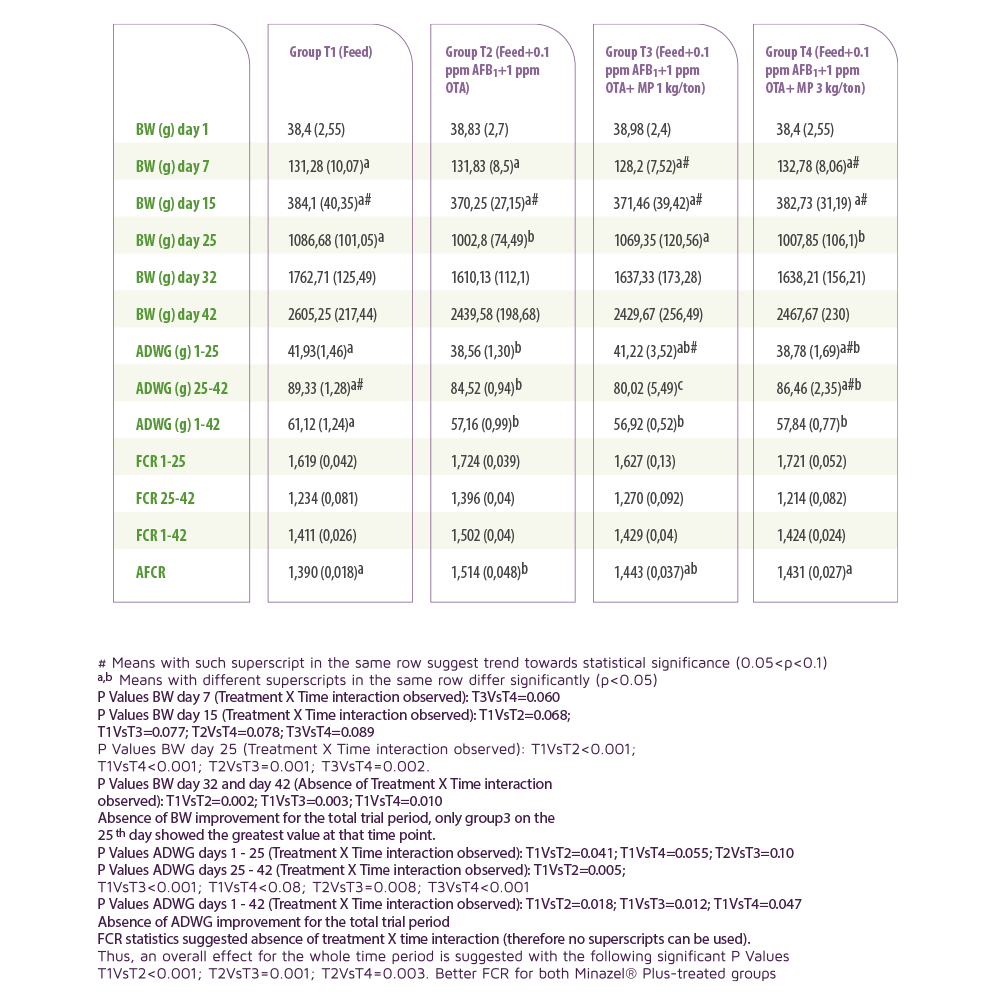
The data presented in Table 1 showed the better FCR for Minazel® Plus treated groups.
Absence of ADWG improvement was recorded for the total trial period.
A significant improvement of AFCR was observed in the T4 group when compared with T2 group, whilst comparison of AFCR between T3 and T2 was only marginally not significant.
Table 1. Performance Parameters. Values expressed as means (standard deviation in parentheses).
 The data in Table 2 showed a possible liver-protective effect of the test product in the T4 group against mycotoxin-induced liver injury as suggested by differences observed in total proteins and ALT levels.
The data in Table 2 showed a possible liver-protective effect of the test product in the T4 group against mycotoxin-induced liver injury as suggested by differences observed in total proteins and ALT levels.
Table 2. Performance Parameters. Values expressed as means (standard deviation in parentheses).
![]() 1st Blood Sampling
1st Blood Sampling

![]() 2nd Blood sampling
2nd Blood sampling

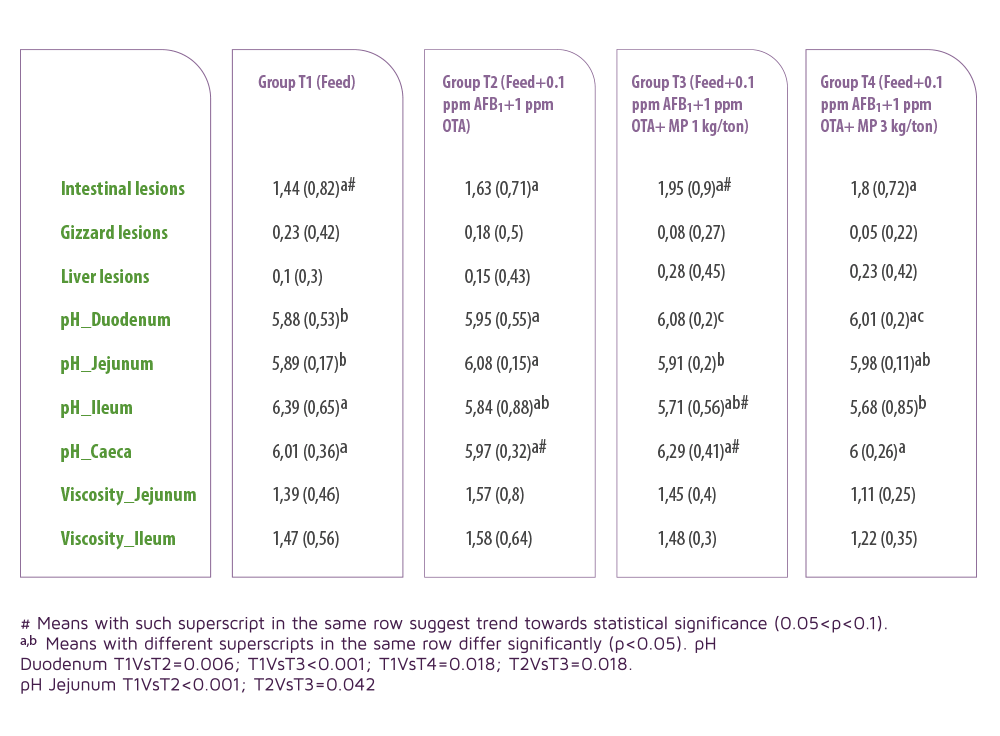
 A significant increase in pH in duodenum and a lower pH at the jejunum was observed in the T3 group.
A significant increase in pH in duodenum and a lower pH at the jejunum was observed in the T3 group.
⇰ This reduction of the pH may inhibit the growth of acid sensitive bacteria, such as Salmonella spp., Campylobacter spp. and Clostridium perfringens.

Table 3. Macroscopic lesions and intestinal findings. Values expressed as means (standard deviation in parentheses).








 Micotoxicosis prevention
Micotoxicosis prevention