Sabry El-khodary
Professor of Internal Medicine and vice dean for post graduate studies and research affair, Faculty of Veterinary Medicine, Mansoura University, Egypt
Ruminant animals are less vulnerable to the adverse effects caused by mycotoxins in comparison to monogastric animals.
⇰ This is based on the hypothesis that the microbiota residing in the rumen can degrade and inactivate mycotoxins, thus protecting the animal (Fink-Gremmels, 2008a).
![]() However, mycotoxins that are capable of resisting degradation in the rumen can definitely cause clinical symptoms of intoxication.
However, mycotoxins that are capable of resisting degradation in the rumen can definitely cause clinical symptoms of intoxication.
Dairy cows may be subjected to a varying number of mycotoxins, originating from different feed materials, such as roughage and concentrates (Smith, 2014).
Aflatoxins (AF) produced by Aspergillus species, Fumonisins (FUM), Zearalenone (ZEN), T-2 toxin (T-2), and Deoxynivalenol (DON) produced by Fusarium species, and Ochratoxin A (OTA) produced by Aspergillus and Penicillium species are well-known mycotoxins due to their agricultural importance (Changwa et al., 2018; Njobeh et al., 2012).

![]() Due to a pre-existing negative energy balance, cows in the transition period are particularly sensitive to exposure to feed contaminated with mould, fungal spores, and mycotoxins (Donkor et al., 2016).
Due to a pre-existing negative energy balance, cows in the transition period are particularly sensitive to exposure to feed contaminated with mould, fungal spores, and mycotoxins (Donkor et al., 2016).
Contamination of dairy feed with mycotoxins has been frequently reported, which poses a serious constraint to animal health and productivity.
Mycotoxicosis pathogenesis
Mycotoxins affect animals through different mechanisms and several factors influence the impact they can have, including:
- ⇰ Type of mycotoxin
- ⇰ Animal species
- ⇰ Age of the animal
- ⇰ Level and duration of exposure to mycotoxins
- (Pier et al., 1980)
Some mycotoxins target specific organs such as the liver or the kidneys (Elgioushy et al., 2020). However, environmental conditions, animal health status, and other stresses also play a role in the negative effects of mycotoxins (Tola and Kebede, 2016).
Aspergillus fumigatus is one of the main mycotoxigenic fungus infecting forages under warm conditions (Alonso et al., 2015; Frisvad et al., 2009; Storm et al., 2008).
It produces more than 226 potentially bioactive secondary metabolites (Frisvad et al., 2009).
DON and other Trichothecenes, such as T-2 and HT-2 toxins, Diacetoxyscirpenol (DAS) and Nivalenol (NIV), are mycotoxins produced by Fusarium, and they are suspected to be implicated in farm animal disease outbreak (Antonissen et al., 2014).
Penicillium roqueforti and P. paneum produce several secondary metabolites with immunosuppressive, antibacterial, and other toxicological effects in animals (Gallo et al., 2015; O’Brien et al., 2006).

The starting point for mycotoxins to affect the health and productivity of the animals is the induction of oxidative stress, hypoxia, decreased phagocytosis, and immunosuppression (Oswald et al., 2005).
![]() For example, gliotoxin, an immunosuppressant, has been detected in animals infected with A. fumigatus with a subsequent increase in its virulence (Sutton et al., 1996), and aflatoxins have been found to suppress inflammatory cytokines in cattle (Kurtz and Czuprynski, 1992).
For example, gliotoxin, an immunosuppressant, has been detected in animals infected with A. fumigatus with a subsequent increase in its virulence (Sutton et al., 1996), and aflatoxins have been found to suppress inflammatory cytokines in cattle (Kurtz and Czuprynski, 1992).
 Mycotoxins can negatively affect intestinal integrity, which acts as a physical barrier preventing the passage of pathogens and toxins to the circulatory system. They also increase gut permeability and reduce intestinal cell viability (Awad et al., 2017; Gao et al., 2020).
Mycotoxins can negatively affect intestinal integrity, which acts as a physical barrier preventing the passage of pathogens and toxins to the circulatory system. They also increase gut permeability and reduce intestinal cell viability (Awad et al., 2017; Gao et al., 2020). Mycotoxins affect the gut microbiota by causing an imbalance in the bacterial population.
Mycotoxins affect the gut microbiota by causing an imbalance in the bacterial population.
They decrease the beneficial bacterial populations and increase the pathogenic bacterial populations. Therefore, due to the forementioned pathways, varying degrees of clinical illness can occur (Guerre, 2020).
For example, in addition to its effects on reproduction, Zearalenone and Its metabolites can cause changes in the rumen ecosystem soon after intake, including ruminal eukaryotes (Hartinger et al., 2023).
 Moreover, mycotoxins cause damage to the liver tissue and, consequently, influence its detoxification capacity (Średnicka et al., 2021).
Moreover, mycotoxins cause damage to the liver tissue and, consequently, influence its detoxification capacity (Średnicka et al., 2021).
Fumonisins act by altering sphingolipid biosynthesis, leading to the accumulation of sphinganine and causing toxicity (Mathur et al., 2001).
They also induce the formation of neutrophil extracellular traps (NETs), increases reactive oxygen species (ROS) levels, and decreases SOD and CAT activities. Concurrently, exposure to fumonisins results in concentration-dependent phosphorylation of ERK-1/2 and p38 in neutrophils (Wang et al., 2020).
 Mycotoxins produced by Fusarium spp. and Aspergillus spp. in dairy cow diets can have a negative influence on somatic cell count, blood parameters and immunity (Jovaišienė et al., 2016).
Mycotoxins produced by Fusarium spp. and Aspergillus spp. in dairy cow diets can have a negative influence on somatic cell count, blood parameters and immunity (Jovaišienė et al., 2016).
Additionally, primary antibody response to ovalbumin immunization and serum sodium concentrations and osmolality are higher in cows fed a diet contaminated with Fusarium mycotoxins, but neutrophil phagocytosis was depressed (Korosteleva et al., 2009).
Citrinin is a nephrotoxic mycotoxin produced by several species of the genera Aspergillus and Penicillium (Chain, 2012), but it can also occur also as an undesirable contaminant in Monascus ruber fermentation products and concurrent occurrence of OTA in food or feed materials (Bouslimi et al., 2008).
![]() Field experiments have suggested that cows fed citrinin-contaminated diets show signs of pruritus, pyrexia, and hemorrhagic syndrome, as well as fever, diarrhea, and uremia (Lloyd, 1980).
Field experiments have suggested that cows fed citrinin-contaminated diets show signs of pruritus, pyrexia, and hemorrhagic syndrome, as well as fever, diarrhea, and uremia (Lloyd, 1980).
![]() Mycotoxins may potentially affect reproductive function in cattle (Caloni et al., 2009).
Mycotoxins may potentially affect reproductive function in cattle (Caloni et al., 2009).
ZEARALENONE
![]() A controlled study with non-lactating cows fed 500 mg of Zearalenone (calculated dietary concentrations of approximately 25 ppm zearalenone) showed that corpora lutea were smaller in treated cows.
A controlled study with non-lactating cows fed 500 mg of Zearalenone (calculated dietary concentrations of approximately 25 ppm zearalenone) showed that corpora lutea were smaller in treated cows.
![]() Zearalenone intake through dietary feed, even when below the threshold contamination level permitted in Japan, may affect the ovarian antral follicle populations, but not the fertility, of post-partum cows (Fushimi et al., 2015).
Zearalenone intake through dietary feed, even when below the threshold contamination level permitted in Japan, may affect the ovarian antral follicle populations, but not the fertility, of post-partum cows (Fushimi et al., 2015).
![]() It has been found that Zearalenone contamination is also detrimental to oocyte quality in heifers (Silva et al., 2021).
It has been found that Zearalenone contamination is also detrimental to oocyte quality in heifers (Silva et al., 2021).

Clinical symptoms associated with exposure to mycotoxins
Mycotoxins, alkaloids, certain lactones, and coumarins, as well as eicosanoic carboxylic esters, are not degraded (at least, not completely) by the rumen microbiota and can produce clinical signs (Fink-Gremmels, 2008b).
Specific toxins affect specific organs or tissues such as the liver, kidney, oral and gastric mucosa, brain, or reproductive tract (Fink-Gremmels, 2008b).
Mycotoxins can cause acute episodes when animals consume critical quantities.
⇰ In acute mycotoxicosis, the signs of disease are often marked and directly referable to the affected target organs.
However, at lower concentrations (chronic exposure), the effects of mycotoxins are more variable.
⇰ They reduce the growth rate of young animals and some interfere with the mechanisms of disease resistance and impair immune responsiveness, making the animals more susceptible to infections (Pier et al., 1980).
In these circumstances is frequent to see chronic issues, including a higher incidence of disease, poor reproductive performance, or suboptimal milk production (Whitlow y Hagler Jr, 2010).
Symptoms associated with exposure to Aflatoxins
Based on field experience, the main clinical findings of aflatoxicosis in ruminant animals are:
- ⇰ Sudden death (Image 1)
- ⇰ Diarrhea (Image 2)
- ⇰ Anorexia
- ⇰ Rough coat

Image 1. Sudden death in a cow due to aflatoxicosis.

Image 2. A cow with signs of diarrhea due to aflatoxicosis.
![]() An outbreak of aflatoxicosis in cattle demonstrated that depression and inappetence were the predominant clinical findings (Elgioushy et al., 2020).
An outbreak of aflatoxicosis in cattle demonstrated that depression and inappetence were the predominant clinical findings (Elgioushy et al., 2020).
![]() Van Halderen et al. (1989) described a field outbreak mortality of 7/25 calves fed rations containing maize with 11.790 μg of Aflatoxins/kg.
Van Halderen et al. (1989) described a field outbreak mortality of 7/25 calves fed rations containing maize with 11.790 μg of Aflatoxins/kg.
⇰ Clinical signs included loss in body mass, rough coat, diarrhea, and rectal prolapse.
![]() In Australia, acute aflatoxicosis that caused mortality of 12/90 drought-stricken calves fed on peanut hay containing of 2230 μg AF/kg was reported (McKenzie et al., 1981).
In Australia, acute aflatoxicosis that caused mortality of 12/90 drought-stricken calves fed on peanut hay containing of 2230 μg AF/kg was reported (McKenzie et al., 1981).
![]() In Pakistan, 45 field cases of aflatoxicosis on a local farm in Okara were recorded (Sohooa et al., 2015).
In Pakistan, 45 field cases of aflatoxicosis on a local farm in Okara were recorded (Sohooa et al., 2015).
⇰ The cows were fed corn-rich forage with 33.500 μg AF/kg and the clinical signs observed were anorexia, depression, photosensitization, and diarrhea, which resulted in the death of 15 animals.
![]() significant decrease in milk production in cattle fed 13 mg AFB1/day for 7 days was reported (Applebaum et al., 1982).
significant decrease in milk production in cattle fed 13 mg AFB1/day for 7 days was reported (Applebaum et al., 1982).
![]() A numerical drop in milk yield in cows fed 75 μg/kg Dry Matter Intake (DMI) (1725 μg/head per day) for 5 days was also reported (Ogunade et al., 2016) and (Jiang et al., 2018).
A numerical drop in milk yield in cows fed 75 μg/kg Dry Matter Intake (DMI) (1725 μg/head per day) for 5 days was also reported (Ogunade et al., 2016) and (Jiang et al., 2018).
![]() In another study (Sulzberger et al., 2017), depression in milk yield and feed conversion at 100 μg AFB1/kg of DMI was recorded.
In another study (Sulzberger et al., 2017), depression in milk yield and feed conversion at 100 μg AFB1/kg of DMI was recorded.
![]() In contrast, some studies on low levels of aflatoxins have shown a non-significant decrease in milk production (Kutz et al., 2009; Masoero et al., 2007; Sumantri et al., 2012).
In contrast, some studies on low levels of aflatoxins have shown a non-significant decrease in milk production (Kutz et al., 2009; Masoero et al., 2007; Sumantri et al., 2012).
![]() For example, reduced gas production, dry matter digestibility, and ammonia-N concentrations caused by AFB1 in vitro have been reported (Mojtahedi et al., 2013).
For example, reduced gas production, dry matter digestibility, and ammonia-N concentrations caused by AFB1 in vitro have been reported (Mojtahedi et al., 2013).
![]() In another study, AFB1 reduced ammonia-N and volatile fatty acid (VFA) concentrations without reducing dry matter digestibility or affecting VFA patterns (Jiang et al., 2012).
In another study, AFB1 reduced ammonia-N and volatile fatty acid (VFA) concentrations without reducing dry matter digestibility or affecting VFA patterns (Jiang et al., 2012).
Symptoms associated with exposure to Deoxynivalenol
DON has been found to affect ruminal fermentation and can cause reduced milk yield (Whitlow and Hagler, 2010).
![]() In a study on the effect of a diet contaminated with DON in non-lactating Holstein dairy cows fed at a dose of 1.5 mg DON/kg and 6.4 mg DON/kg of feed for 6 weeks, there was a slight decline in feed consumption following the change from the low dose (1.5 mg DON/kg) to the high dose (6.4 mg DON/kg) (Trenholm et al., 1985).
In a study on the effect of a diet contaminated with DON in non-lactating Holstein dairy cows fed at a dose of 1.5 mg DON/kg and 6.4 mg DON/kg of feed for 6 weeks, there was a slight decline in feed consumption following the change from the low dose (1.5 mg DON/kg) to the high dose (6.4 mg DON/kg) (Trenholm et al., 1985).
![]() In calves, severe liver failure in 2–3-month-old calves with no functioning rumen induced by 1.13 mg DON/kg feed was reported (Valgaeren et al., 2019).
In calves, severe liver failure in 2–3-month-old calves with no functioning rumen induced by 1.13 mg DON/kg feed was reported (Valgaeren et al., 2019).

Symptoms associated with exposure to Fumonisins
Ruminants are more resistant to fumonisin toxicity than monogastric animals (Mathur et al., 2001).
![]() However, feeding trials with 75 mg FB1/ kg, 94 mg FB1/kg, and 105 mg FB1/ kg for 14 days, 253 days, and 31 days, respectively, have been reported to cause reduced milk yield, reduced feed intake, hepatotoxicity, nephrotoxicity, and reproduction problems (Richard et al., 1996).
However, feeding trials with 75 mg FB1/ kg, 94 mg FB1/kg, and 105 mg FB1/ kg for 14 days, 253 days, and 31 days, respectively, have been reported to cause reduced milk yield, reduced feed intake, hepatotoxicity, nephrotoxicity, and reproduction problems (Richard et al., 1996).
![]() Experimental intravenous administration of 1 mg FB1/kg to calves for 7 days caused lethargy, loss of appetite, hepatotoxicity, and nephrotoxicity (Mathur et al., 2001).
Experimental intravenous administration of 1 mg FB1/kg to calves for 7 days caused lethargy, loss of appetite, hepatotoxicity, and nephrotoxicity (Mathur et al., 2001).
![]() In a study with milk-fed male Holstein calves aged 7 to 14 days treated daily with 1 mg FB1/kg (iv) until euthanasia on day 7, treated calves were lethargic and had decreased appetite from day 4 onward (Mathur et al., 2001).
In a study with milk-fed male Holstein calves aged 7 to 14 days treated daily with 1 mg FB1/kg (iv) until euthanasia on day 7, treated calves were lethargic and had decreased appetite from day 4 onward (Mathur et al., 2001).
Symptoms associated with exposure to Ochratoxins
Ochratoxicosis is rarely reported in cattle, which is attributed to the ability of the rumen microbiota to easily degrade OTA to non-toxic forms as demonstrated (Richard et al., 1996).
![]() However, anorexia, diarrhea, difficulty in rising, and cessation of milk production with recovery on the 4th day in cattle fed a high single dose of OTA (13.3 mg DON/ kg) has been recorded experimentally, whereas in the field low doses (0.2 mg DON/kg, 0.75 mg DON/kg, and 1.66 mg DON/kg) for 5 days produced no clinical disease (Ribelin et al., 1978).
However, anorexia, diarrhea, difficulty in rising, and cessation of milk production with recovery on the 4th day in cattle fed a high single dose of OTA (13.3 mg DON/ kg) has been recorded experimentally, whereas in the field low doses (0.2 mg DON/kg, 0.75 mg DON/kg, and 1.66 mg DON/kg) for 5 days produced no clinical disease (Ribelin et al., 1978).
Symptoms associated with exposure to T-2 toxin
In dairy cattle, T-2 toxin has been associated with hemorrhagic gastroenteritis (Whitlow and Hagler Jr, 2010), feed refusal, and gastrointestinal lesions (Weaver et al., 1980).
![]() Weaver et al. (1980) reported severe depression, hindquarter ataxia, knuckling of the rear feet, listlessness, and anorexia in a calf fed 0.6 mg T-2 toxin/kg for seven consecutive days.
Weaver et al. (1980) reported severe depression, hindquarter ataxia, knuckling of the rear feet, listlessness, and anorexia in a calf fed 0.6 mg T-2 toxin/kg for seven consecutive days.
![]() Reduction in milk yield and the absence of estrus have also been associated with T-2 (Whitlow and Hagler, 2010).
Reduction in milk yield and the absence of estrus have also been associated with T-2 (Whitlow and Hagler, 2010).
Symptoms associated with exposure to Zearalenone
![]() Early abortion in cattle feeding on hay containing 10 mg ZEN/kg has been recorded (Kallela y Ettala, 1984).
Early abortion in cattle feeding on hay containing 10 mg ZEN/kg has been recorded (Kallela y Ettala, 1984).
![]() Abnormal estrus cycle, vaginitis, behavioral estrus in gestating animals, mammary development in prepuberal heifers, and sterility have also been reported in cattle fed 1.5 mg ZEN/kg feed (Gupta, 2012).
Abnormal estrus cycle, vaginitis, behavioral estrus in gestating animals, mammary development in prepuberal heifers, and sterility have also been reported in cattle fed 1.5 mg ZEN/kg feed (Gupta, 2012).
![]() Experimental studies using 500 mg and 250 mg of 99% purified Zearalenone in a gelatin capsule administered orally to lactating dairy cattle and virgin heifers, respectively, showed no effects except for depression in the conception rate in the virgin heifers (Weaver et al., 1986a, b).
Experimental studies using 500 mg and 250 mg of 99% purified Zearalenone in a gelatin capsule administered orally to lactating dairy cattle and virgin heifers, respectively, showed no effects except for depression in the conception rate in the virgin heifers (Weaver et al., 1986a, b).
Symptoms associated with other fungal related infections
![]() In an experimental study, Diplodiosis, a type of neuromycotoxicosis that mainly affects cattle, characterized by ataxia, paresis and paralysis, was induced in 13 heads of cattle, 16 sheep and 3 goats by dosing them with Diplodia maydis (Kellerman et al., 1985).
In an experimental study, Diplodiosis, a type of neuromycotoxicosis that mainly affects cattle, characterized by ataxia, paresis and paralysis, was induced in 13 heads of cattle, 16 sheep and 3 goats by dosing them with Diplodia maydis (Kellerman et al., 1985).
![]() In Australia, Acute Bovine Liver Disease (ABLD) was recorded in 45 naturally affected cattle from 13 outbreaks occurring from 2010 to 2019.
In Australia, Acute Bovine Liver Disease (ABLD) was recorded in 45 naturally affected cattle from 13 outbreaks occurring from 2010 to 2019.
⇰ Clinical signs commonly included a combination of mild photosensitization, progressive neurologic signs, and hypogalactia, which preceded death in less than 48 hours (Manthorpe et al., 2021).
Lesions associated with exposure to mycotoxins
Aflatoxin-induced lesions
Based on field experience, the gall bladder, liver, and kidneys are the most commonly affected organs in cases of aflatoxicosis (Images 3, 4 and 5).
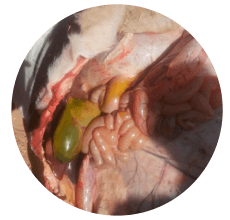
Image 3. Enlarged gall bladder in cow dead affected by aflatoxicosis.

Image 4. Congested kidney in cattle afflicted with aflatoxicosis.
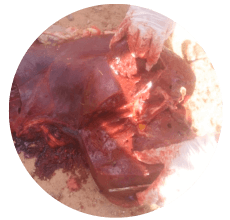
Image 5. Liver with necrotic areas from a cow with aflatoxicosis.
Acute aflatoxin toxicity has been found to provoke major signs of liver lesions, leading to congestion and bleeding (Pier, 1992; Pier and Richard, 1992).
Aflatoxicosis also causes fatty acid accumulation in the liver, kidneys, and heart, and may be responsible for encephalopathies and oedemas (Pfohl-Leszkowicz, 2000).
However, chronic aflatoxicosis is more common and, in this case, the liver is the main target.
Aflatoxins act as DNA intercalating agents, binding to guanine bases and leading to cell death or tumor formation (Pfohl-Leszkowicz, 2000).
Fumonisin-induced lesions
![]() Oral administration of a diet containing FB1 (2.36 mg FB1/kg/day increased to 3.54 mg FB1/kg/day) to calves for 239 to 253 days showed elevated sphinganine/sphingosine ratios with mild hepatocellular morphological changes accompanied by mild bile duct epithelial changes (Baker y Rottinghaus, 1999).
Oral administration of a diet containing FB1 (2.36 mg FB1/kg/day increased to 3.54 mg FB1/kg/day) to calves for 239 to 253 days showed elevated sphinganine/sphingosine ratios with mild hepatocellular morphological changes accompanied by mild bile duct epithelial changes (Baker y Rottinghaus, 1999).
![]() Sphinganine and sphingosine concentrations in liver, kidney, lung, heart, and skeletal muscle were increased in milk-fed male Holstein calves aged 7 to 14 days treated with at 1 mg/kg (iv), daily until euthanized on day 7.
Sphinganine and sphingosine concentrations in liver, kidney, lung, heart, and skeletal muscle were increased in milk-fed male Holstein calves aged 7 to 14 days treated with at 1 mg/kg (iv), daily until euthanized on day 7.
⇰ In this case, the hepatic lesions were characterized by disorganized hepatic cords, varying severity of hepatocyte apoptosis, hepatocyte proliferation, and proliferation of bile ductular cells.
⇰ Additionally, renal lesions consisted of vacuolar changes, apoptosis, karyomegaly, and proliferation of proximal renal tubular cells, as well as dilation of proximal renal tubules (Mathur et al., 2001).
![]() Moreover, significant increases in serum liver enzyme activity with mild microscopic liver lesions and eventual impairment of lymphocyte blastogenesis were described in two calves fed at the highest Fumonisin level (Osweiler et al., 1993).
Moreover, significant increases in serum liver enzyme activity with mild microscopic liver lesions and eventual impairment of lymphocyte blastogenesis were described in two calves fed at the highest Fumonisin level (Osweiler et al., 1993).
Lesions associated with other fungal related infections
![]() In an experimental study dosing cattle, sheep, and goats with Diplodia maydis, an extensive laminar subcortical status of spongiosis was evident in the cerebrum and cerebellum of a sheep that had been long paralyzed and a steer that had permanent locomotory disturbance (Kellerman et al., 1985).
In an experimental study dosing cattle, sheep, and goats with Diplodia maydis, an extensive laminar subcortical status of spongiosis was evident in the cerebrum and cerebellum of a sheep that had been long paralyzed and a steer that had permanent locomotory disturbance (Kellerman et al., 1985).
![]() In Australia, in cases of Acute Bovine Liver Disease (ABLD) the main histological lesions were severe periportal hepatocellular coagulative necrosis and erythrocyte pooling, which often extended to massive necrosis.
In Australia, in cases of Acute Bovine Liver Disease (ABLD) the main histological lesions were severe periportal hepatocellular coagulative necrosis and erythrocyte pooling, which often extended to massive necrosis.
REFERENCES
Albonico, M., Schutz, L.F., Caloni, F., Cortinovis, C., Spicer, L.J., 2017. In vitro effects of the Fusarium mycotoxins fumonisin B1 and beauvericin on bovine granulosa cell proliferation and steroid production. Toxicon 128, 38-45.
Alonso, V., Díaz Vergara, L., Aminahuel, C., Pereyra, C., Pena, G., Torres, A., Dalcero, A., Cavaglieri, L., 2015. Physiological behaviour of gliotoxigenic Aspergillus fumigatus sensu stricto isolated from maize silage under simulated environmental conditions. Food Additives & Contaminants: Part A 32, 236-244.
Antonissen, G., Martel, A., Pasmans, F., Ducatelle, R., Verbrugghe, E., Vandenbroucke, V., Li, S., Haesebrouck, F., Van Immerseel, F., Croubels, S., 2014. The impact of Fusarium mycotoxins on human and animal host susceptibility to infectious diseases. Toxins 6, 430-452.
Applebaum, R.S., Brackett, R.E., Wiseman, D.W., Marth, E.H., 1982. Responses of dairy cows to dietary aflatoxin: Feed intake and yield, toxin content, and quality of milk of cows treated with pure and impure aflatoxin. Journal of Dairy Science 65, 1503-1508.
Awad, W.A., Hess, C., Hess, M., 2017. Enteric pathogens and their toxin-induced disruption of the intestinal barrier through alteration of tight junctions in chickens. Toxins 9, 60.
Baker, D.C., Rottinghaus, G.E., 1999. Chronic experimental fumonisin intoxication of calves. Journal of Veterinary Diagnostic Investigation 11, 289-292.
Bouslimi, A., Bouaziz, C., Ayed-Boussema, I., Hassen, W., Bacha, H., 2008. Individual and combined effects of ochratoxin A and citrinin on viability and DNA fragmentation in cultured Vero cells and on chromosome aberrations in mice bone marrow cells. Toxicology 251, 1-7.
Caloni, F., Ranzenigo, G., Cremonesi, F., Spicer, L.J., 2009. Effects of a trichothecene, T-2 toxin, on proliferation and steroid production by porcine granulosa cells. Toxicon 54, 337-344.
Chain, E.P.o.C.i.t.F., 2012. Scientific Opinion on the risks for public and animal health related to the presence of citrinin in food and feed. EFSA Journal 10, 2605.
Changwa, R., Abia, W., Msagati, T., Nyoni, H., Ndleve, K., Njobeh, P., 2018. Multi-mycotoxin occurrence in dairy cattle feeds from the gauteng province of South Africa: A pilot study using UHPLC-QTOF-MS/MS. Toxins 10, 294.
Donkor, O., Ramchandran, L., Vasiljevic, T., 2016. Techniques 8 and Control of Mycotoxins Detection, Quantification in Dairy Products. Microbial Toxins in Dairy Products 7, 201.
Elgioushy, M.M., Elgaml, S.A., El-Adl, M.M., Hegazy, A.M., Hashish, E.A., 2020. Aflatoxicosis in cattle: clinical findings and biochemical alterations. Environmental Science and Pollution Research 27, 35526-35534.
Fink-Gremmels, J., 2008a. The role of mycotoxins in the health and performance of dairy cows. Vet J 176, 84-92.
Fink-Gremmels, J., 2008b. The role of mycotoxins in the health and performance of dairy cows. The Veterinary Journal 176, 84-92.
Frisvad, J.C., Rank, C., Nielsen, K.F., Larsen, T.O., 2009. Metabolomics of Aspergillus fumigatus. Medical Mycology 47, S53-S71.
Fushimi, Y., Takagi, M., Monniaux, D., Uno, S., Kokushi, E., Shinya, U., Kawashima, C., Otoi, T., Deguchi, E., Fink-Gremmels, J., 2015. Effects of Dietary Contamination by Zearalenone and Its Metabolites on Serum Anti-Müllerian Hormone: Impact on the Reproductive Performance of Breeding Cows. Reprod Domest Anim 50, 834-839.
Gallo, A., Giuberti, G., Bertuzzi, T., Moschini, M., Masoero, F., 2015. Study of the effects of PR toxin, mycophenolic acid and roquefortine C on in vitro gas production parameters and their stability in the rumen environment. The Journal of Agricultural Science 153, 163-176.
Gao, Y., Meng, L., Liu, H., Wang, J., Zheng, N., 2020. The compromised intestinal barrier induced by mycotoxins. Toxins 12, 619.
Gonçalves, B.L., Corassin, C.H., Oliveira, C.A.F.d., 2015. Mycotoxicoses in dairy cattle: a review. Asian Journal of Animal and Veterinary Advances 10, 752-760.
Guerre, P., 2020. Mycotoxin and gut microbiota interactions. Toxins 12, 769.
Gupta, R.C., 2012. Veterinary toxicology: basic and clinical principles. Academic press.
Hartinger, T., Kröger, I., Neubauer, V., Faas, J., Doupovec, B., Schatzmayr, D., Zebeli, Q., 2023. Zearalenone and Its Emerging Metabolites Promptly Aff ect the Rumen Microbiota in Holstein Cows Fed a Forage-Rich Diet. Toxins (Basel) 15.
Jiang, Y., Ogunade, I., Kim, D., Li, X., Pech-Cervantes, A., Arriola, K., Oliveira, A., Driver, J., Ferraretto, L., Staples, C., 2018. Effect of adding clay with or without a Saccharomyces cerevisiae fermentation product on the health and performance of lactating dairy cows challenged with dietary aflatoxin B1. Journal of dairy science 101, 3008-3020.




 Micotoxicosis prevention
Micotoxicosis prevention