Dr. Marco Iammarino is a food technologist and chemical surveyor with over two decades of experience in food quality and safety. Since 2002, he has worked as a Researcher at the Istituto Zooprofilattico Sperimentale della Puglia e della Basilicata (Italy), where he develops and validates analytical methods for the detection of food additives, contaminants, veterinary drugs, pesticides, and mycotoxins.
Dr. Iammarino is an active member of several national and international working groups, including EFSA, ISO, CEN, and UNI, and serves on the editorial boards of prominent journals such as Journal of Food Composition and Analysis and Frontiers in Nutrition. His contribution to scientific publishing has been widely recognized, having been ranked among the top 1 % peer reviewers by Clarivate Publons.
While his scientific output includes more than 300 publications, a significant portion of his recent work focuses on the emerging risks posed by multi-mycotoxin contamination. His 2025 research proposal addresses the complexity of mycotoxin mixtures in foods, exploring their structural and functional interactions, potential synergistic toxicity, and the implications for food safety policy. In this interview, we discuss the importance of shifting from single-compound risk models to integrated approaches, the analytical challenges involved in detecting mycotoxin cocktails, and the future of regulatory frameworks in an era of increasingly complex food systems.

You have dedicated over 20 years to analytical chemistry applied to food safety. How did you first become interested in mycotoxins, and how has your focus evolved over time?
Dealing with food safety from a chemical perspective, environmental contaminants were among the first subjects of my investigations, particularly mycotoxins and their possible presence in products of animal origin.
 In this regard, I worked extensively on the determination of total aflatoxins, fumonisins, deoxynivalenol, ochratoxin A, and others in food and animal feed, all within the framework of official activities carried out by the health authority (namely, the Experimental Zooprophylactic Institute of Puglia and Basilicata), where I conduct my research activity.
In this regard, I worked extensively on the determination of total aflatoxins, fumonisins, deoxynivalenol, ochratoxin A, and others in food and animal feed, all within the framework of official activities carried out by the health authority (namely, the Experimental Zooprophylactic Institute of Puglia and Basilicata), where I conduct my research activity.
Over the years, we developed a series of research projects on mycotoxins funded by the Italian Ministry of Health.
During these projects, several analytical methods for the determination of these mycotoxins in both food for human consumption and livestock products were developed, validated, and subsequently accredited for the official control of food and animal feed.
Regarding the evolution of analytical techniques, this research began about twenty years ago.
At that time, many determinations were carried out using ELISA methods, which were gradually replaced by more advanced technologies.
 As far as mycotoxins are concerned, HPLC has found many applications, particularly when combined with fluorimetric detection, which allows very high sensitivity levels to be achieved, sometimes comparable to those of mass spectrometry (e.g., for total aflatoxins, fumonisins, ochratoxin A, zearalenone, etc.).
As far as mycotoxins are concerned, HPLC has found many applications, particularly when combined with fluorimetric detection, which allows very high sensitivity levels to be achieved, sometimes comparable to those of mass spectrometry (e.g., for total aflatoxins, fumonisins, ochratoxin A, zearalenone, etc.).
 In recent years, however, mass spectrometry has gained prominence, as it enables multi-residue analysis of different mycotoxins in a single chromatographic run. This feature significantly simplifies the routine work of food and animal feed control laboratories, especially when confirmatory analyses are required.
In recent years, however, mass spectrometry has gained prominence, as it enables multi-residue analysis of different mycotoxins in a single chromatographic run. This feature significantly simplifies the routine work of food and animal feed control laboratories, especially when confirmatory analyses are required.


Your recent research proposal addresses a crucial but often overlooked reality: that mycotoxins are frequently present as mixtures. What motivated you to focus on this topic, and what do you see as the main scientific and regulatory gaps in this area?
This is a relatively new aspect in food safety.
 The concept was introduced by EFSA in its first official documents in 2013 concerning so-called “chemical mixtures”, namely combinations of different contaminants in the same food product.
The concept was introduced by EFSA in its first official documents in 2013 concerning so-called “chemical mixtures”, namely combinations of different contaminants in the same food product.
In practice, the risk assessment of multiple chemicals is conducted using a tiered approach that includes:
- ⇒ Exposure assessment
- ⇒ Hazard assessment
- ⇒ Risk characterization
For instance, let us consider the official control of chemical contaminants in a milk sample.
 Regulation (EU) 2023/915 provides a list of contaminants to be determined (aflatoxin M1, lead, dioxins, and PCBs), each with a specific maximum level.
Regulation (EU) 2023/915 provides a list of contaminants to be determined (aflatoxin M1, lead, dioxins, and PCBs), each with a specific maximum level.
Usually, during official controls, the determination of a specific contaminant (or class of contaminants) is carried out.
 The question is: what about a sample in which all listed contaminants are present at levels just below the legal limit?
The question is: what about a sample in which all listed contaminants are present at levels just below the legal limit?
 The sample is, of course, compliant, but what about the possible cumulative effect of such a cocktail of contaminants?
The sample is, of course, compliant, but what about the possible cumulative effect of such a cocktail of contaminants?
In this regard, the first studies were developed on pesticides, given the high number of such compounds and their simultaneous use and, in some cases, cumulative effects were reported.
However, apart from a few sporadic cases, such studies struggle to gain traction, mainly because they are highly challenging and proper financial support is often difficult to secure.

Can you explain the potential risks associated with synergistic or antagonistic interactions between mycotoxins? Are there any examples or findings that particularly highlight the importance of studying these combinations?
With reference to the possible presence of different contaminants in the same food product, mycotoxins certainly represent a particularly interesting category of compounds.
Indeed, certain groups of mycotoxins — such as those produced by the same fungus under similar temperature and humidity conditions and on the same raw material — may be simultaneously present as contaminants in the final product.
 This is a situation that can also be observed in other cases, such as pesticides or other groups of environmental contaminants.
This is a situation that can also be observed in other cases, such as pesticides or other groups of environmental contaminants.
 In the case of mycotoxins, it is reasonable to consider the possibility of a synergistic toxic effect exerted by these compounds.
In the case of mycotoxins, it is reasonable to consider the possibility of a synergistic toxic effect exerted by these compounds.This topic is currently under investigation, and the scientific literature reports several studies demonstrating the co-occurrence of different mycotoxins (mainly zearalenone, deoxynivalenol, aflatoxins, and fumonisins) in both animal feed and grains such as wheat and oats.
Potentially, the presence of multiple mycotoxins could lead to synergistic effects. However, some studies have shown that the interaction may be merely additive — as reported for fumonisins, aflatoxins, ochratoxin A, deoxynivalenol, and diacetoxyscirpenol — or even antagonistic, as in the case of deoxynivalenol and ochratoxin A.
 These effects have been described in studies carried out on poultry feed samples.
These effects have been described in studies carried out on poultry feed samples.


From an analytical perspective, what are the main challenges in detecting and quantifying multiple mycotoxins simultaneously? How are emerging technologies like high-resolution mass spectrometry helping to overcome these hurdles?
 Until a few years ago, these determinations were performed on single mycotoxins using techniques such as ELISA and/ or liquid or thin-layer chromatography, which allowed the identification and quantification of individual mycotoxins.
Until a few years ago, these determinations were performed on single mycotoxins using techniques such as ELISA and/ or liquid or thin-layer chromatography, which allowed the identification and quantification of individual mycotoxins.
 However, the introduction of mass spectrometry gradually has made it possible to determine multiple mycotoxins simultaneously within the same chromatographic run, with excellent analytical sensitivity and selectivity.
However, the introduction of mass spectrometry gradually has made it possible to determine multiple mycotoxins simultaneously within the same chromatographic run, with excellent analytical sensitivity and selectivity.
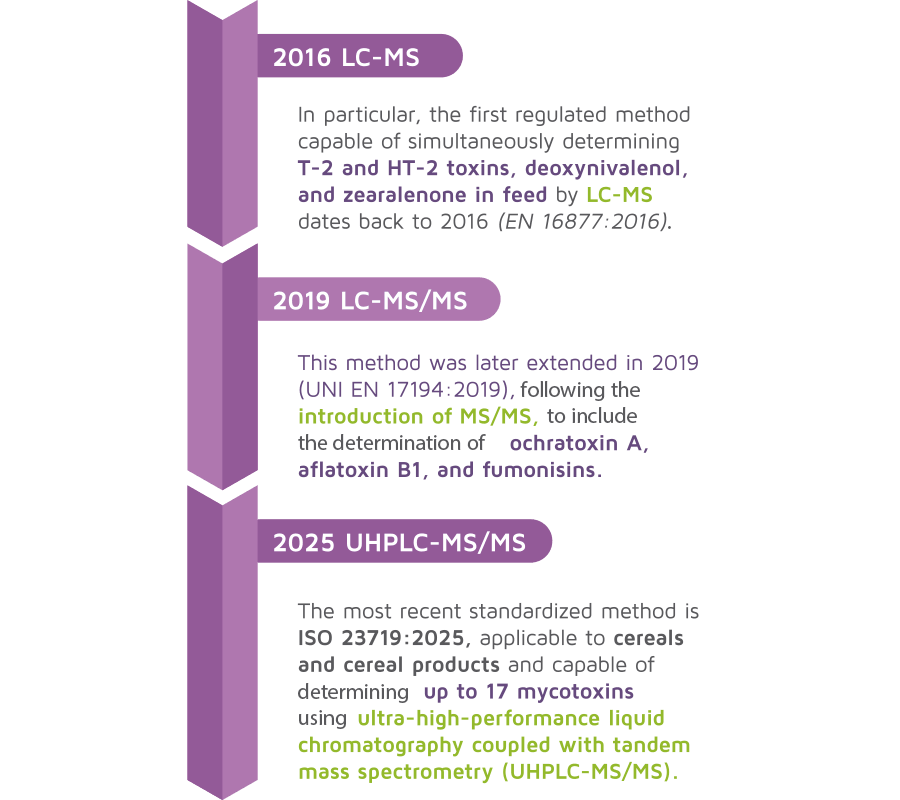
These methods are undoubtedly very effective in the context of official control of food and animal feed, particularly from a Green Chemistry perspective.
 Specifically, point 8 of the well-known AGREE pictogram assigns a high score when a large number of analytes can be detected within the same chromatographic run.
Specifically, point 8 of the well-known AGREE pictogram assigns a high score when a large number of analytes can be detected within the same chromatographic run.
If we wish to be particularly rigorous, some challenges may arise when applying multi-residue determinations of mycotoxins by UPLC-MS/MS or high-resolution mass spectrometry (HRMS) to specific samples that require complex sample preparation steps (e.g., during extraction or purification).
 In such cases, the approach may not fully guarantee the determination of all target mycotoxins as expected, due to potential losses in sensitivity and/or the presence of interfering compounds. However, this remains a relatively minor limitation.
In such cases, the approach may not fully guarantee the determination of all target mycotoxins as expected, due to potential losses in sensitivity and/or the presence of interfering compounds. However, this remains a relatively minor limitation.


In one of your studies, you report five years of official control of mycotoxins in animal feed. What trends have you observed in terms of contamination levels, and how do these results inform risk management in the feed industry?
The study we conducted was carried out over a five-year period and focused on the evaluation of different mycotoxins, namely aflatoxin B1, zearalenone, deoxynivalenol, ochratoxin A, fumonisins, and T-2/HT-2 toxins.
Overall, 722 samples of animal feed were collected and analysed using fully validated and accredited analytical methods based on both ELISA (used as a screening method) and HPLC-FLD (as a confirmatory technique).
 Fourteen samples showed mycotoxin concentrations exceeding the related maximum residue limits (MRLs).
Fourteen samples showed mycotoxin concentrations exceeding the related maximum residue limits (MRLs). Five, four, and five non-compliant samples were identified for deoxynivalenol (DON), aflatoxins (AF), and zearalenone (ZEN), respectively.
Five, four, and five non-compliant samples were identified for deoxynivalenol (DON), aflatoxins (AF), and zearalenone (ZEN), respectively. Corn was identified as the most frequently occurring raw material in these non-compliant samples.
Corn was identified as the most frequently occurring raw material in these non-compliant samples.
Particular attention was paid to possible correlations between mycotoxin type and animal susceptibility.
 The study highlighted that swine is the most affected species, as it is highly susceptible to zearalenone, deoxynivalenol, and T-2/HT-2 toxins, and the mean concentrations detected during this survey were close to the respective legal limits.
The study highlighted that swine is the most affected species, as it is highly susceptible to zearalenone, deoxynivalenol, and T-2/HT-2 toxins, and the mean concentrations detected during this survey were close to the respective legal limits.
 Interesting findings were also obtained regarding rabbits and poultry in relation to T-2/HT-2 toxin contamination in feed. The mean contamination levels recorded were close to half of the legal limit, and these two animal species are particularly susceptible to these toxins.
Interesting findings were also obtained regarding rabbits and poultry in relation to T-2/HT-2 toxin contamination in feed. The mean contamination levels recorded were close to half of the legal limit, and these two animal species are particularly susceptible to these toxins.
Considering these findings, special precautions should be applied during the manufacturing and storage of feed for swine, rabbits, and poultry to achieve an overall reduction in contamination by certain mycotoxins, particularly zearalenone, deoxynivalenol, and T-2/HT-2 toxins.

You have also worked on method validation for ELISA and UPLC/FLD as screening and confirmatory techniques. How do you balance the need for speed and sensitivity in routine monitoring programs?
For laboratories required to analyse a high number of samples within established deadlines, as is the case for official control laboratories, the possibility of using analytical methods capable of processing many samples in a short time while delivering reliable results is essential.
In this context, ELISA approaches are particularly useful, as they allow a large number of samples to be analysed rapidly and, in many cases, minimize the risk of “false negative” results.
The analytical sensitivity of these methods often meets regulatory requirements in terms of limits of quantification and established legal limits.
In this regard, we published a specific study in which we carried out a point-to-point comparison between ELISA and UPLC/FLD for the determination of T-2/HT-2 toxins in different types of cereals.
 This allowed us to evaluate the most significant advantages and limitations of each approach, thereby supporting laboratory managers in selecting the most appropriate method.
This allowed us to evaluate the most significant advantages and limitations of each approach, thereby supporting laboratory managers in selecting the most appropriate method.
Certain characteristics, such as method selectivity and sensitivity, were higher for instrumental techniques, particularly when fluorometric detection was coupled with solid-phase immunoaffinity extraction.
 This combination ensures the absence of interfering signals and very low limits of quantification.
This combination ensures the absence of interfering signals and very low limits of quantification.
Regarding accuracy performance:
 Method trueness (evaluated as recovery %) was comparable between the two approaches.
Method trueness (evaluated as recovery %) was comparable between the two approaches. Method precision (CV %) was slightly better for ELISA due to the simpler sample preparation procedure compared with UPLC/FLD, which involves additional steps that may reduce repeatability.
Method precision (CV %) was slightly better for ELISA due to the simpler sample preparation procedure compared with UPLC/FLD, which involves additional steps that may reduce repeatability.
Beyond validation parameters, the two approaches were also compared by analyzing 100 cereal samples using both methods.
 The concentrations quantified by both techniques (27 samples with concentrations above the ELISA limit of 75 ppb) were compared using linear regression analysis.
The concentrations quantified by both techniques (27 samples with concentrations above the ELISA limit of 75 ppb) were compared using linear regression analysis.
A good correlation (r = 0.9056; n = 27) was observed, demonstrating that the concentrations obtained with the two approaches overlap when a reasonable measurement uncertainty is taken into account.
Finally, regarding the possibility of obtaining “false positive” results with ELISA, the proportion of such responses was 2 %, which does not significantly impact the routine activity of a food control laboratory.

When it comes to mycotoxin mixtures, how well do you think current regulatory frameworks reflect the real risks consumers face? Is there any movement at the European or international level toward adapting legislation to account for co-contamination?
As mentioned before, the issue of simultaneous exposure to multiple contaminants concerns several categories of chemical compounds.
 Among these, mycotoxins certainly represent a highly significant group, as the possibility that several mycotoxins may be present simultaneously in a food product is by no means negligible.
Among these, mycotoxins certainly represent a highly significant group, as the possibility that several mycotoxins may be present simultaneously in a food product is by no means negligible.
Indeed, the conditions that favor the development of these compounds — in terms of temperature, humidity, and raw material composition — may promote the production of more than one mycotoxin.
From a legislative perspective, and with regard to the possible future enactment of specific regulations, a fundamental role lies with the European Food Safety Authority (EFSA).
The crucial point will be the identification, with the lowest possible margin of uncertainty, of combinations of mycotoxins that can realistically occur simultaneously in specific products or raw materials and that exert a proven synergistic effect.
This would require:
- ⇒ The development of monitoring plans at the European level
- ⇒ The collection of a statistically significant body of data
- ⇒ The performance of toxicological studies to characterize the toxicity of these mixtures
Based on established approaches such as the Margin of Exposure (MOE), it may then become possible to:
 Define specific combinations of mycotoxins
Define specific combinations of mycotoxins Establish corresponding legal limits expressed as the sum of different compounds
Establish corresponding legal limits expressed as the sum of different compounds


Your proposal also emphasizes mechanistic research into the biochemical pathways affected by mycotoxin combinations. Why is this molecularlevel understanding important, and how could it reshape toxicological risk assessment?
This topic clearly falls within the scope of the “One Health” approach, which involves exploring specific issues by considering all physical, environmental, biological, and chemical factors that may influence or modify the overall scenario.
 Regarding mycotoxins, it is evident that the transfer of these toxic compounds from food and feed matrices to humans and animals can be influenced by multiple factors, many of which have a significant impact on human health and animal welfare.
Regarding mycotoxins, it is evident that the transfer of these toxic compounds from food and feed matrices to humans and animals can be influenced by multiple factors, many of which have a significant impact on human health and animal welfare.
 The research proposal particularly focuses on the possible interactions between mycotoxin-producing microorganisms, the mycotoxins themselves, and, once ingested, the way these compounds may be influenced by the intestinal microbiota.
The research proposal particularly focuses on the possible interactions between mycotoxin-producing microorganisms, the mycotoxins themselves, and, once ingested, the way these compounds may be influenced by the intestinal microbiota.
Only a limited number of studies have addressed these aspects, and further investigations are needed to refine risk assessment approaches so that they better reflect real-world exposure scenarios, while also strengthening their relevance from a regulatory perspective.
Historically, both research and regulation have focused on individual mycotoxins, generating valuable information on their toxicological profiles.
However, this approach does not fully reflect the reality that consumers are often exposed to multiple mycotoxins simultaneously.
 These interactions, particularly when synergistic, introduce an additional and still insufficiently addressed layer of complexity in food safety and risk assessment.
These interactions, particularly when synergistic, introduce an additional and still insufficiently addressed layer of complexity in food safety and risk assessment.

Looking ahead, what scientific advances or crossdisciplinary collaborations do you believe will be key to improving how we detect, assess, and regulate multi-mycotoxin risks in food and feed?
Undoubtedly, the issue of mycotoxins in food involves several scientific sectors that are equally important throughout the food supply chain.
Upstream, the topic encompasses agronomic and storage factors related to raw materials, to the extent that FAO and WHO, within the framework of the Codex Alimentarius, have published the “Code of Practice for the Prevention and Reduction of Mycotoxin Contamination in Cereal,” which has been revised over the years.
At the next stage, the presence of mycotoxins also depends on the specific processing applied to raw materials.
Therefore, close collaboration between food technologists and analytical chemists is essential to evaluate the actual presence and levels of these compounds.
At this point in the food supply chain, it would also be valuable to assess how innovative product stabilization technologies — such as high-pressure processing, irradiation, and other emerging approaches — may contribute to decontamination and long-term stabilization.
 Further downstream, in the context of risk assessment, the contribution of toxicologists with expertise in biology and chemistry is fundamental to thoroughly characterize the toxicity of these compounds and their possible mixtures.
Further downstream, in the context of risk assessment, the contribution of toxicologists with expertise in biology and chemistry is fundamental to thoroughly characterize the toxicity of these compounds and their possible mixtures.
 The role of medical doctors and microbiologists is also highly relevant in investigating potential interactions between mycotoxins and the microbiota following ingestion.
The role of medical doctors and microbiologists is also highly relevant in investigating potential interactions between mycotoxins and the microbiota following ingestion.




 Micotoxicosis prevention
Micotoxicosis prevention